Sepsis ImmunoScore®
First-ever FDA authorized AI sepsis diagnostic tool with powerful diagnostic and predictive capabilities
De Novo FDA Authorization (April 2024)
We believe acute care should be tailored to the individual biology of each patient.
We enable clinicians to see people differently with new tools that reveal deep biological insights.
See People Differently
See beyond...
symptoms
See beyond...
age
See beyond...
notes
See beyond...
gender
See beyond...
diagnosis
See beyond...
weight
See beyond...
protocols
See beyond...
race
A Next Gen Precision Medicine Tool for Acute Care
Sepsis ImmunoScore assists providers in combatting sepsis, which kills more people than all cancers combined.1
Sepsis ImmunoScore enables faster treatment decisions, improved outcomes, quality metrics, and hospital financials.
![]()
FDA De Novo Authorized
![]()
Built on Biological Data, Trained on Thousands of Patients
![]()
22 Holistic Parameters
![]()
Artificial Intelligence to Augment Clinician Decision Making
![]()
A Diagnostic integrated Into the Workflow: Not an Alert System
![]()
Complementary, Not Competitive, to Alert Systems
Sepsis ImmunoScore results in four risk categories that are associated with:

Chances of Sepsis within 24 hours
In-Hospital Mortality
Hospital Length of Stay

ICU Transfer Within 24 Hours

Vasopressors Used Within 24 Hours
Mechanical Ventilation Within 24 Hours
Building Trust with Clinicians
Most artificial intelligence solutions for sepsis are a black box.
Sepsis ImmunoScore – Transparency to enable true clinician-AI partnership
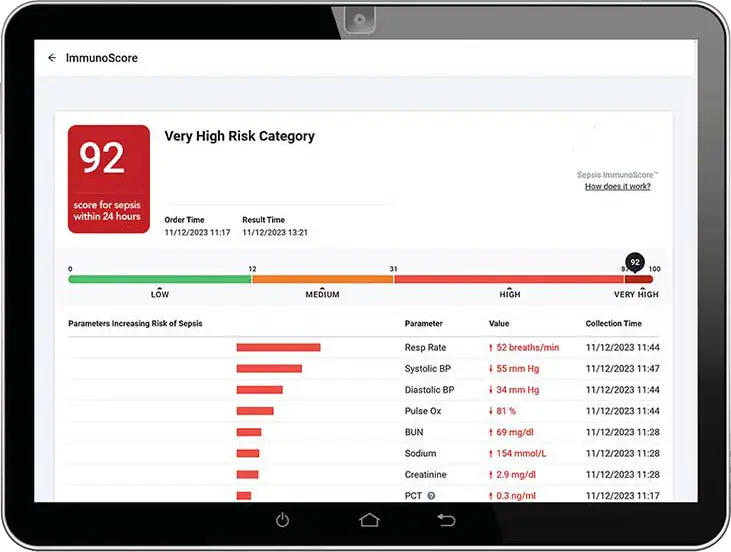
Each Sepsis ImmunoScore result is accompanied by an intuitive display that directly explains how each of the 22 patient parameters either increased or decreased the risk of sepsis for that individual patient. In a human factors study2 with >30 clinicians, 100% agreed that the graphical interface de-mystified AI and built trust in the Sepsis ImmunoScore.
1. Rudd, K. E. et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. The Lancet 395, 200–211 (2020).
2. Human factors study performed for FDA De Novo Approval.